Call us now :+8613788933411
Vorapaxar Sulfa Intermediate
MOQ : 1 Ton
Vorapaxar Sulfa Intermediate Specification
- Storage
- Room Temperature
- Other Names
- Vorapaxar Sulfa Intermediate
- CAS No
- 900161-13-9
- Grade
- Medicine Grade
- Application
- Pharmaceutical Industry
- Form
- Liquid
Vorapaxar Sulfa Intermediate Trade Information
- Minimum Order Quantity
- 1 Ton
- Delivery Time
- 10 Days
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
About Vorapaxar Sulfa Intermediate
Chemvon Biotechnology is one of the leading high-technology manufacturer in the field of pharmaceutical and fine chemical industry.
From origins as a research group in technology service, chemvon has progressed into an integrated company with a kilo lab and a pilot plant offering a range of APIs, NCEs and some organic intermediates.
Quality:Adopting the leading technology to assure the high quality.
Price:Offering the best price among the suppliers in China.
Rich material and energy make us control the cost effective
Afrer-sale service:Chemvon will take charge of everything if there is any question after the sale.
Certificates: We can offer ISO9001 certificates that you need for importing.
Medical and Industrial Relevance
This intermediate is a key ingredient in the production of Vorapaxar, which is utilized in cardiovascular therapy. Its pharmaceutical-grade quality ensures reliable performance in manufacturing environments, aiding in the creation of life-saving medicines. As a liquid, it allows for easier handling and integration into various synthesis processes within the industry.
Global Distribution and Supply
Distributed and exported worldwide from China, Vorapaxar Sulfa Intermediate is available through manufacturers, suppliers, and traders to pharmaceutical firms across different continents. The controlled supply chain ensures product integrity and consistent availability, making it a dependable choice for large-scale and specialized pharmaceutical applications.
FAQs of Vorapaxar Sulfa Intermediate:
Q: How should Vorapaxar Sulfa Intermediate be stored for optimal quality?
A: Vorapaxar Sulfa Intermediate should be stored at room temperature to maintain stability and prevent degradation, ensuring its pharmaceutical-grade purity and efficacy are preserved until use.Q: What is the main application of Vorapaxar Sulfa Intermediate in the pharmaceutical industry?
A: The primary application of Vorapaxar Sulfa Intermediate is in the synthesis of Vorapaxar, a cardiovascular drug used to reduce the risk of thrombotic events in patients.Q: When is this intermediate typically utilized during the drug manufacturing process?
A: This intermediate is generally used during the chemical synthesis stage of Vorapaxar production, where it serves as a building block enabling the creation of the final active pharmaceutical ingredient.Q: Where is Vorapaxar Sulfa Intermediate predominantly manufactured and exported from?
A: The product is chiefly manufactured, supplied, and exported from China by recognized producers and distributors specializing in pharmaceutical raw materials.Q: What is the process for sourcing Vorapaxar Sulfa Intermediate from China?
A: Pharmaceutical companies and traders can obtain Vorapaxar Sulfa Intermediate by contacting authorized Chinese manufacturers, exporters, or suppliers, ensuring adherence to international trade regulations and quality standards.Q: How is this intermediate used in pharmaceutical formulations?
A: Vorapaxar Sulfa Intermediate is incorporated during the drug synthesis process, contributing essential structural components required for the creation of high-purity Vorapaxar.Q: What are the main benefits of using a medicine-grade liquid intermediate in pharmaceutical production?
A: Employing a liquid, medicine-grade intermediate promotes efficient handling, precise dosing, and consistent quality, supporting dependable manufacturing outcomes and advancing therapeutic research.
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Pharmaceutical Chemicals Category
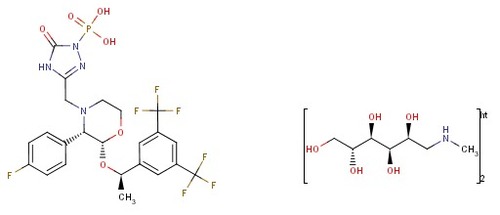
fosaprepitant dimeglumine
Minimum Order Quantity : 1 Ton
CAS No : 265121048
Application : Pharmaceutical Industry
Grade : Medicine Grade
Other Names : Fosaprepitant dimeglumine;DGlucitol, 1deoxy1(methylamino), (3(((2R,3S)2((1R)1(3,5bis(trifluoromethyl)phenyl)ethoxy)3(4fluorophenyl)4 morpholinyl)methyl)2,5dihydro5oxo1H1,2,4triazol1yl)phosphonate (2:1) (salt)
Storage : Freezer
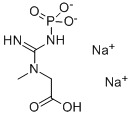
Phosphocreatine disodium salt
Minimum Order Quantity : 1 , , Ton
CAS No : 19333654
Application : Pharmaceutical Industry
Grade : Medicine Grade
Storage : Room Temperature
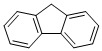
Fluorene Chemical
Minimum Order Quantity : 1 , , Ton
CAS No : 86737
Application : Pharmaceutical Industry
Grade : Medicine Grade
Other Names : Fluorene Chemical
Storage : Room Temperature
Benzonitrile Onitrile
Minimum Order Quantity : 1 Ton
CAS No : 20099892
Application : Pharmaceutical Industry
Grade : Medicine Grade
Other Names : Benzonitrile Onitrile
Storage : Room Temperature

 Send Inquiry
Send Inquiry



 Send Inquiry
Send Inquiry