Call us now :+8613788933411
Calcifediol Chemical
MOQ : 1 Ton
Calcifediol Chemical Specification
- Molecular Formula
- C27H44O2
- Storage
- Freezer
- Place of Origin
- China
- Molecular Weight
- 400.64 Grams (g)
- Other Names
- CALCIDIOL;25-HYDROXYVITAMIN D3;25-HYDROXYCHOLECALCIFEROL;VITAMIN D3, 25-HYDROXY-;25-HYDROXYVITAMIN D3 WHITE CRYSTALLINE SOLID 985;25-Hydroxyvitamin D3
- CAS No
- 19356-17-3
- Type
- Vitamins
- Grade
- Medicine Grade
- Usage
- Calcium supplement
- Purity
- 98%min
- Appearance
- white solid
- Application
- Pharmaceutical Industry
- Form
- Solid
Calcifediol Chemical Trade Information
- Minimum Order Quantity
- 1 Ton
- FOB Port
- Shanghai
- Payment Terms
- Letter of Credit (L/C), Letter of Credit at Sight (Sight L/C), Telegraphic Transfer (T/T), Paypal
- Supply Ability
- 1000 Tons Per Week
- Delivery Time
- 15 Days
- Sample Available
- Yes
- Sample Policy
- Contact us for information regarding our sample policy
- Packaging Details
- foil/bag/bottle
- Main Export Market(s)
- Australia, North America, Eastern Europe, South America, Western Europe, Africa, Central America, Middle East, Asia
About Calcifediol Chemical
We offer Calcifediol with the following product specification:
Features:
- High purity
- Free sample available
- Lower price
- Experienced service team
- Fast delivery
- Considerate after-sale service
Looking for the ideal CAS no.19356-17-3 manufacturer & supplier? we are in the occupation of providing a superior quality range of Calcifediol to our most valued client. Each product are professionally under sophisticated detection equipment of a reliable quality assurance. We are China origin factory of High purity Calcifediol. If you have any question, please dont hesitate to contact us.
High Purity and Quality
Our Calcifediol boasts a minimum purity of 98%, ensuring consistency and safety for pharmaceutical applications. As a medicine-grade compound, it is manufactured with meticulous quality control, providing reliable results for vitamin D3 supplementation.
Pharmaceutical Applications
Calcifediol is primarily utilized in the pharmaceutical industry for its efficacy as a calcium supplement and vitamin D3 source. Its high purity and stability make it ideal for manufacturing medicines that treat and prevent vitamin D deficiencies and related illnesses.
Proper Storage and Handling
To maintain the integrity of Calcifediol, it should be stored in a freezer under solid form. Proper storage conditions are crucial for preserving potency and preventing degradation, ensuring the compound remains effective during its shelf life.
FAQs of Calcifediol Chemical:
Q: How is Calcifediol typically used in the pharmaceutical industry?
A: Calcifediol is primarily used as a calcium supplement and to treat or prevent vitamin D deficiencies. Its role is vital in producing medications that regulate calcium metabolism and support bone health.Q: What are the benefits of using Calcifediol as a supplement?
A: Calcifediol helps to maintain optimal levels of vitamin D in the body, which is essential for calcium absorption, bone strength, and overall metabolic health. It is especially beneficial for individuals with impaired vitamin D metabolism.Q: Where is Calcifediol sourced from?
A: This compound is manufactured and exported from China, with strict adherence to international quality standards, making it suitable for global supply and distribution.Q: What is the recommended storage condition for Calcifediol?
A: Calcifediol should be stored in a freezer in its solid form. This ensures its stability, preserves its medicinal properties, and extends shelf life.Q: How can Calcifediol be beneficial for pharmaceutical manufacturers?
A: Pharmaceutical manufacturers benefit from its high purity and consistent quality, enabling them to formulate reliable medications for calcium supplementation and vitamin D deficiency treatment.Q: What process ensures the high purity of Calcifediol?
A: The compound undergoes stringent manufacturing and purification processes, followed by rigorous quality control tests to guarantee a purity of at least 98%, suitable for medicine grade applications.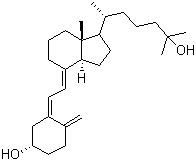
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Pharmaceutical Chemicals Category
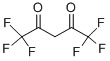
Hexafluoroacetylacetone Chemical
Minimum Order Quantity : 1 Ton
Application : Pharmaceutical Industry
CAS No : 1522221
Grade : Medicine Grade
Molecular Formula : C5H2F6O2
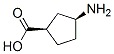
(1R 3S)-3-Aminocyclopentanecarboxylic acid
Minimum Order Quantity : 1 Ton
Application : Pharmaceutical Industry
CAS No : 71830085
Storage : Freezer
Molecular Formula : C6H11NO2
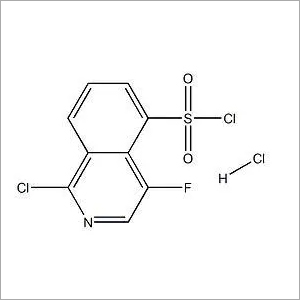
4-Fluoro-5-isoquinolinesulfonyl chloride hydrochloride (1:1)
Minimum Order Quantity : 1 Ton
Application : Pharmaceutical Industry
CAS No : 906820084
Grade : Medicine Grade
Storage : Room Temperature
Molecular Formula : C9H5Cl3FNO2S
Chimmitecan API
Minimum Order Quantity : 1 Ton
Application : Pharmaceutical Industry
CAS No : 185425256
Grade : Medicine Grade
Storage : Room Temperature

 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry