Call us now :+8613788933411
25 hydoxyergocalciferol
MOQ : 1 Ton
25 hydoxyergocalciferol Specification
- Molecular Weight
- 412.65 Grams (g)
- Place of Origin
- China
- Molecular Formula
- C28H44O2
- Other Names
- 25-hydroxyvitamind ;25-hydroxycalciferol;(E,3S,6S)-6-[(1R,3aR,4E,7aS)-4-[(2Z)-2-[(5S)-5-hydroxy-2-methylidene-cyclohexylidene]ethylidene]-7a-methyl-2,3,3a,5,6,7-hexahydro-1H-inden-1-yl]-2,3-dimethyl-hept-4-en-2-ol;25-hydoxyergocalciferol;
- CAS No
- 21343-40-8
- Grade
- Medicine Grade
- Usage
- Supplement vitamin D
- Purity
- 98%min
- Appearance
- white solid
- Application
- Pharmaceutical Industry
- Form
- Solid
25 hydoxyergocalciferol Trade Information
- Minimum Order Quantity
- 1 Ton
- FOB Port
- Shanghai
- Payment Terms
- Letter of Credit (L/C), Letter of Credit at Sight (Sight L/C), Paypal, Telegraphic Transfer (T/T)
- Supply Ability
- 1000 Tons Per Week
- Delivery Time
- 10 Days
- Sample Available
- Yes
- Sample Policy
- Contact us for information regarding our sample policy
- Packaging Details
- foil/bag/bottle
- Main Export Market(s)
- Australia, Africa, Central America, Eastern Europe, Middle East, South America, Western Europe, Asia, North America
About 25 hydoxyergocalciferol
Backed by rich industry experience, we are able to offer superior quality25-hydoxyergocalciferol. Our qualified professionals precisely formulate these drugs from quality chemicals and other ingredients, sourced from the reliable vendors of the industry. These drugs are widely demanded for their effectiveness and non-allergic nature. The drugs we offer are quality examined by our expert members to ensure perfect25-hydoxyergocalciferol. We are able to deliver these products under a specific time-frame at market leading prices.
- CAS NO.: 21343-40-8
- Molecular Formula: C28H44O2
- Molecular Weight: 412.65
Pharmaceutical Applications
25-hydroxyergocalciferol is widely utilized in the pharmaceutical industry as a reliable supplement for vitamin D. Its primary application is in formulations designed to correct or monitor vitamin D deficiencies. The compound is valued for its high purity, ensuring safety and efficacy in clinical use. Pharmaceutical companies rely on this intermediate to produce oral, injectable, and diagnostic vitamin D formulations.
Quality and Standards
With a minimum purity of 98% and meeting medicine-grade specifications, 25-hydroxyergocalciferol assures consistent quality for advanced pharmaceutical needs. Trusted manufacturers and suppliers in China and India adhere to stringent quality controls to maintain these standards, ensuring the product meets international export and medical requirements.
FAQs of 25 hydoxyergocalciferol:
Q: How is 25-hydroxyergocalciferol typically used in pharmaceuticals?
A: 25-hydroxyergocalciferol is used as a supplement and diagnostic agent to assess and manage vitamin D levels in patients, particularly those at risk of deficiency or metabolic disorders related to bone health.Q: What benefits does 25-hydroxyergocalciferol provide?
A: This compound helps support healthy vitamin D status, which is crucial for maintaining bone strength, regulating calcium metabolism, and supporting immune function. In pharmaceutical preparations, it ensures precise dosing and reliable supplementation.Q: When should 25-hydroxyergocalciferol be administered?
A: It is typically administered when a patient shows signs of vitamin D deficiency, is at risk due to specific medical conditions, or needs clinical monitoring of vitamin D levels. Administration should be guided by a healthcare professional.Q: Where is 25-hydroxyergocalciferol manufactured and supplied from?
A: This compound is predominantly produced in China and India, where it is available through various manufacturers, distributors, exporters, and suppliers specializing in pharmaceutical-grade materials.Q: What is the process for ensuring the purity and quality of 25-hydroxyergocalciferol?
A: Manufacturers employ rigorous quality control procedures, including advanced purification and analytical testing, to ensure that the product achieves a minimum purity of 98% and meets medicine-grade standards for pharmaceutical use.Q: How should 25-hydroxyergocalciferol be stored and handled?
A: As a solid white compound, it should be stored in a cool, dry place, away from direct sunlight and moisture. Proper handling practices in accordance with pharmaceutical guidelines are advisable to maintain its efficacy and prevent contamination.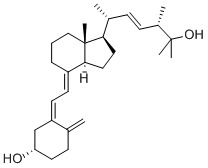
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Pharmaceutical Chemicals Category
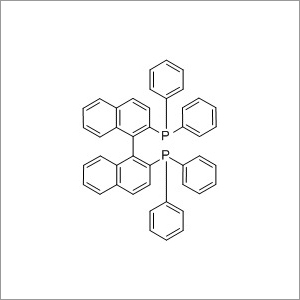
R BINAP
Minimum Order Quantity : 1 Ton
CAS No : 76189554
Storage : Freezer
Grade : Medicine Grade
Form : Solid
Molecular Formula : C44H32P2
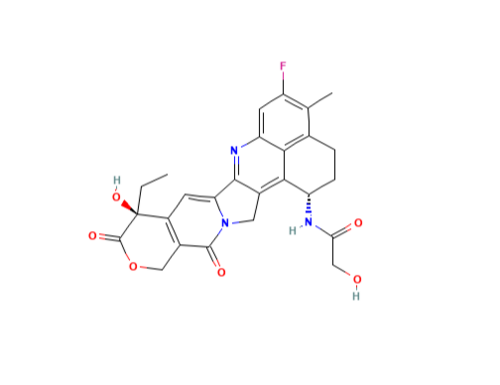
DX-8951 derivative
Minimum Order Quantity : 1 Gram
CAS No : 1599440331
Storage : Room Temperature
Grade : Tech Grade
Form : Liquid
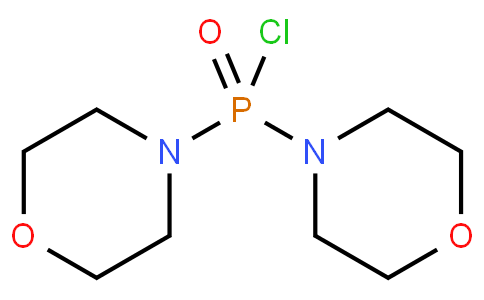
DiMorpholinophosphinyl Chloride
Minimum Order Quantity : 1 Ton
CAS No : 7264906
Storage : Room Temperature
Grade : Medicine Grade
Form : Powder
Molecular Formula : :C8H16ClN2O3P
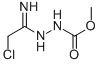
Methyl Z 2 chloro 1 hydrazino ethylidenecarbamate
Minimum Order Quantity : 1 Ton
CAS No : 155742646
Storage : Room Temperature
Grade : Medicine Grade
Form : Solid
Molecular Formula : C4H8CIN3O2

 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry